
The AnalytiChem Laboratory
Technical articles, Industry news and Analysis tips
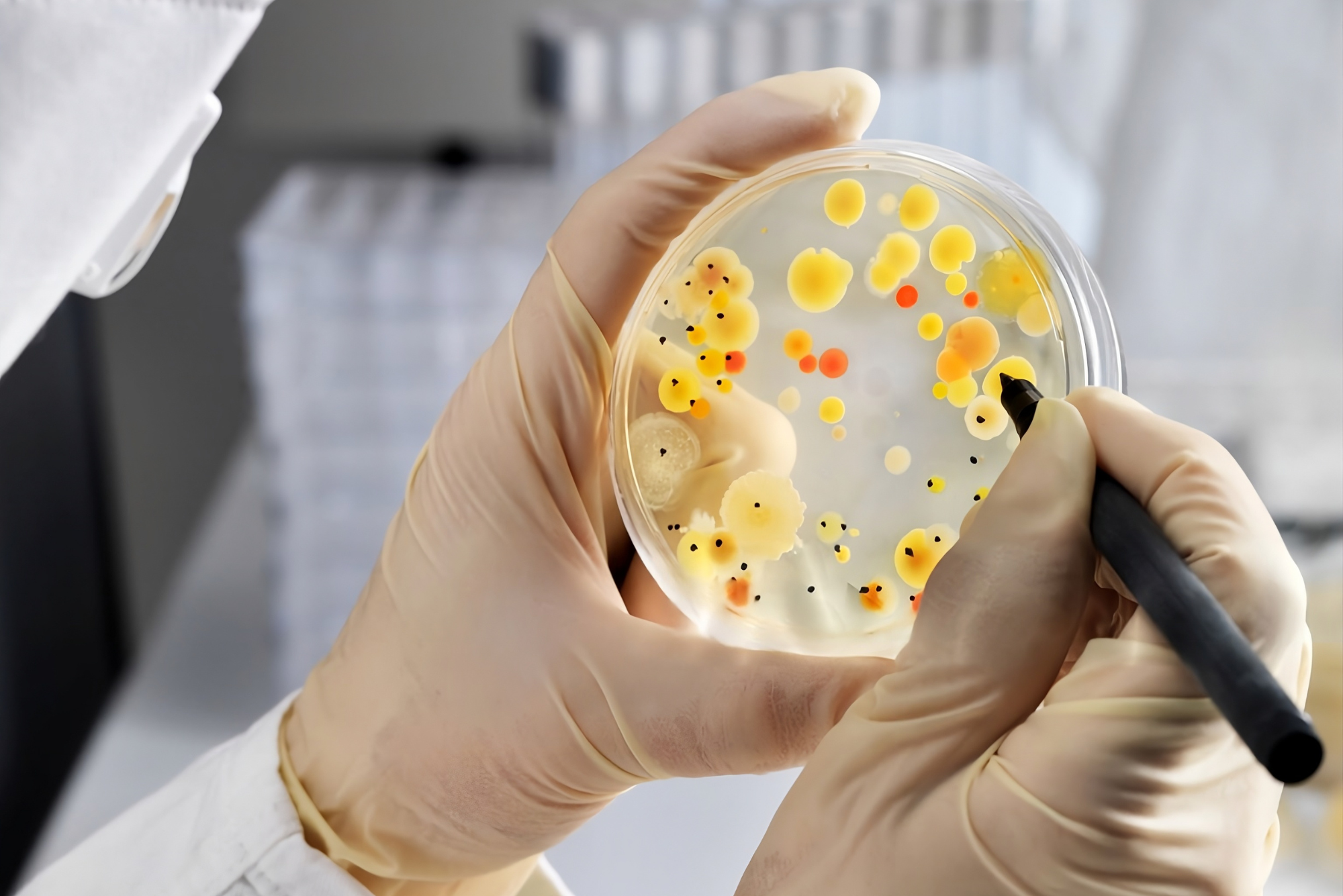
In-House Growth Promotion Testing: Why a Supplier’s Certificate of Analysis is Not Enough
Why your lab's conditions are the final check on media performance.

ISO 11731 Culture Media Selection: BCYE, GVPC, MWY: What to Use and When
Why plate selection determines whether your culture workflow holds up to scrutiny

Should Hospital Pharmacies Review Their Environmental Monitoring Programs Under EU GMP Annex 1?
Why patient risk — not production scale — sets the compliance bar

What Does EU GMP Annex 1 Require for Continuous Environmental Monitoring?
Why continuous monitoring is a risk control mechanism, not a sampling exercise

Annex 1 and GMP: Key Differences Across Manufacturing Scales in Sterile Drug Production
Overview Annex 1 of the EU Guidelines for Good Manufacturing Practice (GMP) provides a unified...
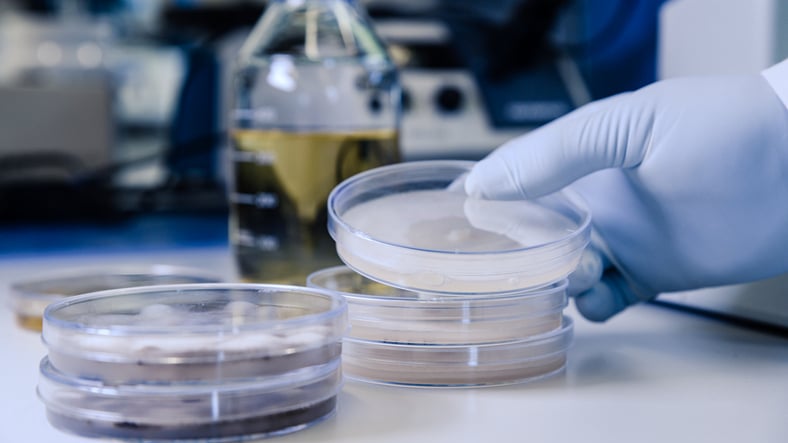
Single-Vent vs. Triple-Vent Microbiology Media Plates: What’s the Difference?
This article explores how single-vent and triple-vent microbiology media plates differ, and how...

Desiccation in Agar Plates: Causes and Prevention
This article covers desiccation of agar in environmental monitoring plates. We'll explain why it...

Why are Some Microbiology Media Autoclaved While Others are Irradiated?
This article explores how sterilization methods are selected for prepared microbiology media: Why...
