
Should Hospital Pharmacies Review Their Environmental Monitoring Programs Under EU GMP Annex 1?
Why patient risk — not production scale — sets the compliance bar
Technical articles, Industry news and Analysis tips

Why patient risk — not production scale — sets the compliance bar

Why continuous monitoring is a risk control mechanism, not a sampling exercise
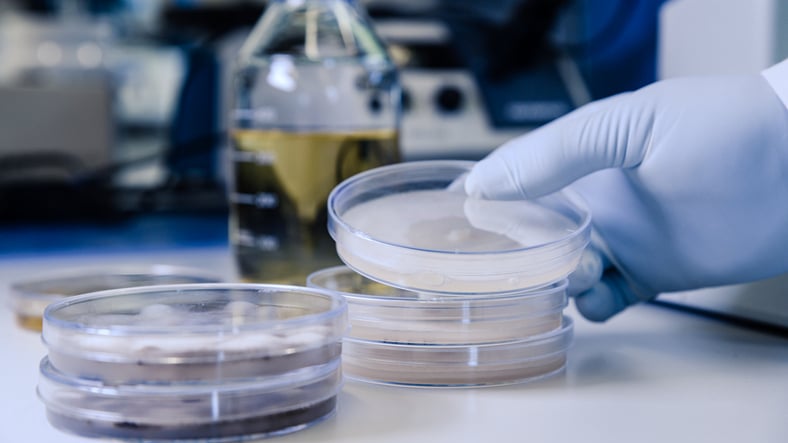
This article explores how single-vent and triple-vent microbiology media plates differ, and how...

Now Available: OREAS 470–474 Clay-Hosted REE CRM Series

This article covers desiccation of agar in environmental monitoring plates. We'll explain why it...

This article explores how sterilization methods are selected for prepared microbiology media: Why...

In this article we’ll cover: Why BSE/TSE remains a concern for users of prepared microbiology...

Selecting a new supplier of prepared microbiology media is a significant decision in pharmaceutical...